GRID2
|
Read other articles:

Konstantin XI Paleolog Dragaš Κωνσταντῖνος ΙΑ' ΠαλαιολόγοςKaisar dari Kekaisaran BizantiumKonstantinus XI PalaiologosBerkuasa6 Januari 1449 – 29 Mei 1453Penobatan6 Januari 1449PendahuluIoannes VIII PalaiologosPenerusJabatan dihapuskanKelahiran(1404-02-08)8 Februari 1404Kematian29 Mei 1453(1453-05-29) (umur 48)AyahManuel II PalaiologosIbuHelena DragašPasanganTheodora ToccoCaterina GattilusioAnakTidak adaAgamaOrtodoks Yunani Konstantinus XI Palaiologos Dragaš...

Stadion River PlateEl Monumental Informasi stadionNama lengkapStadion Monumental Antonio Vespucio LibertiNama lamaEstadio Monumental (1938–1986)PemilikRiver PlateLokasiLokasiBuenos AiresKonstruksiDibuat1936–1938Dibuka25 Mei 1938Direnovasi1978ArsitekJosé AslanHéctor EzcurraData teknisPermukaanRumputKapasitas57,921[1][2][3]Ukuran lapangan105 x 68 mPemakaiArgentinaRiver PlateSunting kotak info • L • BBantuan penggunaan templat ini Stadion Monumental An...

Research collection at University of Kerala This article needs additional citations for verification. Please help improve this article by adding citations to reliable sources. Unsourced material may be challenged and removed.Find sources: Oriental Research Institute & Manuscripts Library – news · newspapers · books · scholar · JSTOR (April 2020) (Learn how and when to remove this template message) Oriental Research Institute The Oriental Research I...
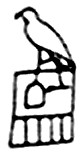
Gambar Nama Horus dalam hieroglif Hor-Aha diperkirakan merupakan raja kedua dari dinasti pertama Mesir. Dia hidup selama abad ke-31 SM. Pada abad ke-32 SM, ayahnya yang bernama Narmer telah menyatukan Mesir Hulu dan Mesir Hilir. Hor-Aha (yang ketika lahir bernama Ity, Hor-Aha adalah gelarnya: 'Horus di antara ilalang') menjadi seorang raja pada usia 30 tahun dan memerintah sampai usia 62 tahun. Menurut legenda, ia dibawa oleh seekor kuda nil, penjelmaan dewa Seth. Cerita lain mengatakan bahwa...

لافتة لمطعم وجبات سريعة في سول عام 2008 يؤكد للزبائن بأنهم يستخدمون لحوم بقر «نظيفة» من أستراليا. جانب من المظاهرات المناوئة لاستيراد لحوم البقر الأمريكية في كوريا الجنوبية بتاريخ 11 مايو عام 2008. مظاهرة حاشدة في وسط سول بتاريخ 6 يونيو عام 2008 احتجاجاً على استئناف استيراد لحوم ا...

Fashion event in New York New York Fashion WeekModel Liu Wen walks the runway modeling fashions by designer Diane von Fürstenberg at New York Fashion Week 2013.GenreClothing and fashion exhibitionsFrequencySemi-annuallyLocation(s)Manhattan, New York City (primarily at Skylight Clarkson Square and Industria)CountryUnited StatesInaugurated1943FounderEleanor Lambert New York Fashion Week (NYFW), held in February and September of each year, is a semi-annual series of events in Manhattan typicall...

Nigerian singer and rapper For the South Korean boy group, see Victon. VictonyBackground informationBirth nameAnthony Ebuka VictorBorn (2001-01-05) 5 January 2001 (age 23)Imo State, NigeriaGenresAfrobeatship hopalternative hip hopR&BOccupation(s)SingersongwriterrapperInstrumentsVocalsYears active2017–presentMusical artist Anthony Ebuka Victor (born 5 January 2001), known professionally as Victony is a Nigerian singer, songwriter and rapper.[1] He is from the Orsu local gov...

Si Biang Kerok CilikGenre Drama Komedi PembuatScreenplay ProductionsSutradara Paulus Pui Ninos Joned Robert Santosa Pemeran Ricky Harun Nina Zatulini Melody Prima Varissa Camelia Indra Birowo Mellissa Grace Desta Negara asalIndonesiaBahasa asliBahasa IndonesiaJmlh. musim1Jmlh. episode149 (daftar episode)ProduksiProduser eksekutifAgus Wijaya AldiProduser Sukhdev Singh Wicky V. Olindo Pengaturan kameraMulti-kameraDurasi60—90 menitRumah produksiScreenplay ProductionsDistributorSurya Citra Med...

この記事は検証可能な参考文献や出典が全く示されていないか、不十分です。出典を追加して記事の信頼性向上にご協力ください。(このテンプレートの使い方)出典検索?: コルク – ニュース · 書籍 · スカラー · CiNii · J-STAGE · NDL · dlib.jp · ジャパンサーチ · TWL(2017年4月) コルクを打ち抜いて作った瓶の栓 コルク(木栓、�...

نيكولاس نكولو (بالفرنسية: Nicolas Nkoulou) معلومات شخصية الميلاد 27 مارس 1990 (العمر 34 سنة)ياوندي الطول 1.84 م (6 قدم 1⁄2 بوصة) مركز اللعب مدافع الجنسية الكاميرون معلومات النادي النادي الحالي أريس تسالونيكي الرقم 3 مسيرة الشباب سنوات فريق Kadji Sports Academy [الإنجليزية] ...

Cemetery in Moscow Kuntsevo CemeteryКунцевское кладбищеDetailsEstablished17th centuryLocationMoscowCountryRussiaCoordinates55°42′28″N 37°25′00″E / 55.70778°N 37.41667°E / 55.70778; 37.41667Size17 hectares (42 acres) The Kuntsevo Cemetery (Russian: Ку́нцевское кла́дбище, romanized: kúntsevkoye kládbishche) is a cemetery servicing Kuntsevo, Moscow. It is located on the bank of the Setun River, to the south of the M...

AnhangueraLokasi di kota São PauloNegara BrasilNegara bagian São PauloWilayahTenggaraKota São PauloZona AdministratifBarat LautSubprefekturPerusBairro Daftar Jardim AnhangüeraJardim BritâniaJardim CleiJardim EscóciaJardim JaraguáMorro DoceVila Chica LuísaVila Jaraguá Luas • Total33,44 km2 (12,91 sq mi)Populasi (2008) • Total58.708 • Kepadatan1.755,39/km2 (4,546,4/sq mi) Distrik Anhanguera adalah nama distrik dalam subp...

American conservative activist and journalist (born 1984) Chris RufoRufo in 2022BornChristopher Ferguson Rufo (1984-08-26) August 26, 1984 (age 39)EducationGeorgetown University (BS)Harvard Extension School (ALM)EmployerManhattan Institute for Policy ResearchKnown forAnti-critical race theory activism Opposition to LGBTQ discussions in classrooms Spouse Suphatra Paravichai (m. 2016)Children3Websitechristopherrufo.com Christopher Ferguson Rufo (born Au...

County in Mississippi, United States County in MississippiTallahatchie CountyCountyTallahatchie County courthouse in SumnerLocation within the U.S. state of MississippiMississippi's location within the U.S.Coordinates: 33°57′N 90°10′W / 33.95°N 90.17°W / 33.95; -90.17Country United StatesState MississippiFoundedDecember 31, 1833Named forChoctaw word roughly meaning “River of rocksSeatCharleston and SumnerLargest cityCharlestonArea • Tota...

Recurring periods of influenza Influenza epidemic redirects here. Not to be confused with Influenza pandemic. This article is about the annual period when flu becomes prevalent. For the TV episode, see Flu Season (Parks and Recreation). This article has multiple issues. Please help improve it or discuss these issues on the talk page. (Learn how and when to remove these template messages) The examples and perspective in this article deal primarily with the United States and do not represent a ...

For the railcar, see Arrow (railcar). Exoatmospheric hypersonic anti-ballistic missile Arrow 3 Arrow 3 test launch, January 2014.TypeExoatmospheric hypersonic anti-ballistic missilePlace of originIsraelService historyUsed byIsraelWars2024 Iranian strikes in IsraelProduction historyDesignerIsrael Aerospace IndustriesManufacturerIsrael Aerospace Industries, BoeingProduced2017–presentSpecificationsEngineTwo-stageOperationalrange2400km[1]Flight ceiling>100kmMaximum spe...

В этом китайском имени фамилия (Хуа) стоит перед личным именем. Для улучшения этой статьи желательно: Найти и оформить в виде сносок ссылки на независимые авторитетные источники, подтверждающие написанное.После исправления проблемы исключите её из списка. Удалите шабло�...

Ukrainian poet and Soviet dissident (born 1939) In this name that follows Eastern Slavic naming customs, the patronymic is Myronovych and the family name is Kalynets. Ihor Mironovych Kalynets, 2007 Ihor Myronovych Kalynets (Ukrainian: Ігор Миронович Калинець; born 1939) is a Ukrainian poet and Soviet dissident. Background Kalynets was born in Khodoriv, the son of an agronomist. His parents upheld Ukrainian cultural traditions. As a child, Kalynets read banned literatu...

此條目需要擴充。 (2017年11月25日)请協助改善这篇條目,更進一步的信息可能會在討論頁或扩充请求中找到。请在擴充條目後將此模板移除。 「對德政策的重述」(Restatement of Policy on Germany )是美國國務卿詹姆斯·弗朗西斯·伯恩斯在1946年9月6日於斯圖加特發表的一篇演講。該演講又稱「希望演講」(Speech of hope),決定了之後美國的對德政策,否定了摩根索計劃抑制德國經�...

Super2 Role Light AircraftType of aircraft National origin United Kingdom Manufacturer ARV Designer Bruce Giddings First flight 11 March 1985 Status Production discontinued Number built about 40 ARV prototype ARV with MidWest AE110, Ecoprop & Trelleborg tyres ARV with Rotax 912 & tundra tyres ARV with Jabiru 2200 The ARV Super2 (Air Recreational Vehicle) is a British two-seat light aircraft with strut-braced shoulder wings and tricycle landing gear.[1] Designed by Bruce Giddi...




