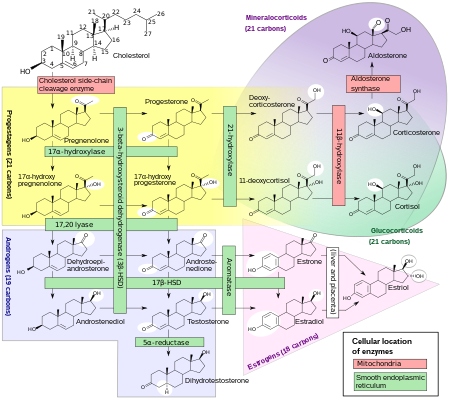
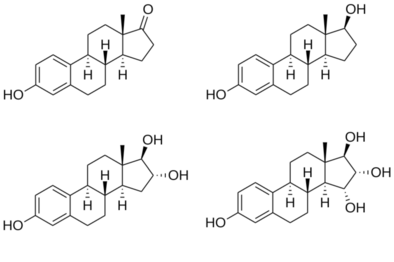
Estriol
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Read other articles:

artikel ini perlu dirapikan agar memenuhi standar Wikipedia. Tidak ada alasan yang diberikan. Silakan kembangkan artikel ini semampu Anda. Merapikan artikel dapat dilakukan dengan wikifikasi atau membagi artikel ke paragraf-paragraf. Jika sudah dirapikan, silakan hapus templat ini. (Pelajari cara dan kapan saatnya untuk menghapus pesan templat ini) PFN Di Jakarta,Indonesia Perusahaan adalah tempat terjadinya kegiatan produksi dan berkumpulnya semua faktor produksi barang dan jasa. Ada perusah...

Port ValeNama lengkapPort Vale Football ClubJulukanThe ValiantsThe ValeThe ValeitesBerdiri1876 / 18791907 (didirikan ulang)StadionVale ParkBurslemStoke-on-Trent(Kapasitas: 15.036)KetuaCarol ShanahanManajerDarren MooreLigaLiga Satu Inggris2022–23ke-18, Liga Satu Inggris Kostum kandang Kostum tandang Kostum ketiga Musim ini Port Vale Football Club adalah sebuah klub sepak bola Inggris yang saat ini bermain di Liga Satu Inggris. Mereka berbasis di Burslem, Staffordshire, salah satu dari enam k...

HeartstringsPoster promosi untuk HeartstringsGenreRomansa, Komedi, DramaDitulis olehLee Myung-sookSutradaraPyo Min-sooPemeranPark Shin-hye Jung Yong-hwa Song Chang-euiLagu penutupYou've Fallen for Me oleh Jung Yong-hwaNegara asalKorea SelatanBahasa asliKoreaJmlh. episode15 +1 spesialProduksiProduser eksekutifChoi Hong-miProduserLee Jin-seok Kim YangDurasi70 menit Rabu dan Kamis pukul 21:55 (WSK)Rumah produksiJS PicturesRilis asliJaringanMBCRilis29 Juni (2011-06-29) –18 Agustus 20...

Election held in Guam Elections in Guam Federal government Presidential straw polls 2008 2012 2016 2020 2024 Presidential caucuses Democratic 2008 2012 2016 2020 2024 Republican 2008 2012 2016 2020 2024 U.S. House of Representatives 2006 2008 2010 2012 2014 2016 2018 2020 2022 2024 Territorial government General elections 1978 1984 1998 2000 2002 2004 2006 2008 2010 2014 2018 2022 Gubernatorial elections 2010 2014 2018 2022 2026 Legislative elections 1931 1933 1946 1950 1952 1954 1956 1960 19...

General Mining Law of 1872Long titleAn Act to promote the Development of the Mining Resources of the United States.Enacted bythe 42nd United States CongressCitationsStatutes at LargeSess. 2, ch. 152, 17 Stat. 91–96Legislative historyIntroduced in the House as H. R. No. 1016 by Aaron A. Sargent (R-CA) on January 15, 1872[1]Committee consideration by House Mines,[1] Senate Mines[2]Passed the House on January 23, 1872 (voice vote[3])Passed th...

Nama ini menggunakan cara penamaan Portugis. Nama keluarga pertama atau maternalnya adalah Bezerra dan nama keluarga kedua atau paternalnya adalah Abbott Galvão. Marcos Galvão Duta Besar Brasil untuk Uni EropaPetahanaMulai menjabat 3 Februari 2019PresidenJair BolsonaroPendahuluEverton Vieira VargasPenggantiPetahanaMenteri Urusan Luar NegeriPelaksana tugasMasa jabatan22 Februari 2017 – 7 Maret 2017PresidenMichel TemerPendahuluJosé SerraPenggantiAloysio Nunes Informasi pri...

Australian actor, director, writer, and comedian Shane JacobsonJacobson in 2013Born (1970-03-18) 18 March 1970 (age 54)Essendon, Victoria, AustraliaNationalityAustralianOccupationsActorentertainerdirectorwritercomedianproducerchief scout of VictoriaYears active1999–presentSpouse Felicity Hunter (m. 2018)Children4[1]Websitewww.shanejacobson.com.au Shane Jacobson (born 18 March 1970) is an Australian actor, director, writer, and comedian, b...

University in Vallam Thanjavur, India This article needs additional citations for verification. Please help improve this article by adding citations to reliable sources. Unsourced material may be challenged and removed.Find sources: Ponnaiyah Ramajayam Institute of Science and Technology – news · newspapers · books · scholar · JSTOR (June 2013) (Learn how and when to remove this message) PRISTMottoQuality and DisciplineTypePrivateEstablished1994Chancel...

Pour les articles homonymes, voir Brecht. Bertolt Brecht Bertolt Brecht en 1954 Données clés Nom de naissance Eugen Berthold Friedrich Brecht Naissance 10 février 1898 Augsbourg, Bavière Empire allemand Décès 14 août 1956 (à 58 ans) Berlin-Est République démocratique allemande Activité principale Dramaturge, metteur en scène, écrivain, poète Distinctions Prix national de la République démocratique allemande (1951) Auteur Langue d’écriture Allemand Œuvres principales ...
2020年夏季奥林匹克运动会波兰代表團波兰国旗IOC編碼POLNOC波蘭奧林匹克委員會網站olimpijski.pl(英文)(波兰文)2020年夏季奥林匹克运动会(東京)2021年7月23日至8月8日(受2019冠状病毒病疫情影响推迟,但仍保留原定名称)運動員206參賽項目24个大项旗手开幕式:帕维尔·科热尼奥夫斯基(游泳)和马娅·沃什乔夫斯卡(自行车)[1]闭幕式:卡罗利娜·纳亚(皮划艇)&#...

Mark Gordon 33º Governatore del WyomingIn caricaInizio mandato7 gennaio 2019 PredecessoreMatt Mead 29° Tesoriere del WyomingDurata mandato1º novembre 2012 –7 gennaio 2019 PredecessoreJoseph Meyer SuccessoreCurt Meier Dati generaliPartito politicoRepubblicano Mark Gordon (New York, 14 marzo 1957) è un politico statunitense, governatore del Wyoming dal 2019. Biografia Si diploma alla Saint Paul's School di Concord e nel 1979 si laurea al Middlebury College a Middle...

Cet article est une ébauche concernant une langue et l’Allemagne. Vous pouvez partager vos connaissances en l’améliorant (comment ?) selon les recommandations des projets correspondants. Frison septentrionalNordfriisk Pays Allemagne Région Schleswig-Holstein Nombre de locuteurs 10 000 (1976)[1] Classification par famille - langues indo-européennes - langues germaniques - langues germaniques occidentales - langues anglo-frisonnes - langues frisonnes - frison septentrional Co...

Lija Il-Kunsill Lokali ta' Ħal LijaLocal councilLija Lambang kebesaranMotto: Suave Fructu Rubeo(With Tasty Fruits I Blossom)Country MaltaIslandMaltaBordersAttard, Balzan, Birkirkara, Iklin, Mosta, NaxxarPemerintahan • MayorIan Castaldi Paris (PN)Luas • Total1,1 km2 (4 sq mi)Populasi (March 2011) • Total2.967 • Kepadatan270/km2 (700/sq mi)DemonimLijan (m), Lijana (f), Lijani (pl)Zona waktuUTC+1 (CET) •&#...

2020 Republican Party presidential primaries ← 2016 February 3 to August 11, 2020 2024 → 2,550 delegate votes (2,443 pledged and 107 unpledged) to the Republican National Convention[1]1,276[1] delegates votes needed to win Candidate Donald Trump Bill Weld Home state Florida[2][a] Massachusetts Delegate count 2,549[1] 1[1] Contests won 56[b][c] 0 Popular vote 18,159,752[1] ...

Wayne Gould (高樂德) (born 3 July 1945 in Hāwera, New Zealand) is a retired Hong Kong judge, most recently known for helping to popularise sudoku puzzles in the United Kingdom, and thereafter in the United States. He pioneered the global success and popularity of the Sudoku puzzle outside Japan where it had been popular for many years. Gould worked as a judge in the criminal courts of Hong Kong. In 1997 he found a sudoku book in a bookstore in Tokyo. Gould then spent 6 years developing a...

American political scandal Further information: Scandals of the Ulysses S. Grant administration Political cartoon by Thomas Nast, March 1876 The Whiskey Ring took place from 1871 to 1876 centering in St. Louis during the presidency of Ulysses S. Grant. The ring was an American scandal, broken in May 1875, involving the diversion of tax revenues in a conspiracy among government agents, politicians, whiskey distillers, and distributors. Whiskey distillers bribed officials from the U. S. Departm...

Grupo Desportivo Estoril PraiaCalcio Canarinhos Segni distintiviUniformi di gara Casa Trasferta Colori sociali Giallo, blu Dati societariCittàEstoril Nazione Portogallo ConfederazioneUEFA Federazione FPF CampionatoPrimeira Liga Fondazione1939 Presidente Alexandre Faria Allenatore Vasco Seabra StadioEstádio António Coimbra da Mota, Estoril(5000 posti) Sito webestorilpraia.pt Palmarès Stagione in corso Si invita a seguire il modello di voce Il Grupo Desportivo Estoril Praia, meglio not...

Football league seasonMacedonian First LeagueSeason2024–25DatesAugust 2024 – May 2025← 2023–24 2025–26 → All statistics correct as of 24 May 2024. The 2024–25 Macedonian First League will be the 33rd season of the Macedonian First Football League, the highest football league of North Macedonia. It will begin in August 2024 and is scheduled to end in May 2025. Struga are the defending champions, having won their second title in 2023–24.[1][2] Promotion and rele...

Girl, InterruptedPoster rilis layar lebarSutradaraJames MangoldProduserDouglas WickCathy KonradSkenarioJames Mangold Lisa Loomer Anna Hamilton PhelanBerdasarkanGirl, Interrupted karya Susanna KaysenPemeran Winona Ryder Angelina Jolie Clea DuVall Brittany Murphy Elisabeth Moss Angela Bettis Jillian Armenante Jared Leto Jeffrey Tambor Vanessa Redgrave Whoopi Goldberg Penata musikMychael DannaSinematograferJack N. GreenPenyuntingKevin TentDistributorColumbia PicturesTanggal rilis 8 Desembe...

Consonne fricative latérale rétroflexe sourde Symbole API ꞎ Numéro API Unicode U+A78E X-SAMPA Kirshenbaum modifier Symboles des fricatives latérales (parmi lesquels le second est la rétroflexe sourde et a été inclus dans Unicode 6.0). La consonne fricative latérale rétroflexe sourde est un son consonantique assez rare. Le symbole utilisé, officiellement dans les extensions de l'alphabet phonétique international, est [ꞎ][1],[2]. Son symbole est, comme toutes les consonnes...