Aluminium phosphate
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Read other articles:
علم المغرب التناسب 2:3 ألوان أحمر أخضر الاعتماد 17 نوفمبر 1915 الاختصاص المغرب التصميم حقل أحمر، تتوسطه نجمة خضراء خماسية المصمم السلطان يوسف علم المغرب الاستعمال شعبي اللواء ألوان 2:3 التناسب 2:3 علم المغرب الاستعمال علم البحرية ألوان 2:3 التناسب 2:3 تعديل مصدري - تعديل ...

Design Research SocietyCompany typeCharityFounded1966HeadquartersUnited KingdomArea servedWorldwideKey peopleJohn Chris Jones, L. Bruce Archer, Nigel CrossWebsitewww.designresearchsociety.org The Design Research Society (DRS), founded in the United Kingdom in 1966,[1][2][3] is an international society for developing and supporting the interests of the design research community. The primary purpose of the DRS, as embodied in its first statement of rules, is to promote �...

Championnats de Belgique de cyclisme sur route Généralités Sport Cyclisme sur route Création 1894 Périodicité Annuelle (juin) Lieu(x) Belgique Disciplines Course en ligne Contre-la-montre Palmarès Tenant du titre Remco Evenepoel Plus titré(s) Tom Steels (4) modifier Les championnats de Belgique de cyclisme sur route sont les championnats nationaux de cyclisme sur route de Belgique, organisés par la Royale ligue vélocipédique belge. Ils ont été disputés pour la première fois en...

James F. ByrnesJames F. Byrnes pada tahun 1941 Gubernur Carolina Selatan ke-104Masa jabatan16 Januari 1951 – 18 Januari 1955WakilGeorge Bell Timmerman, Jr. PendahuluStrom ThurmondPenggantiGeorge Bell Timmerman, Jr.Sekretaris Negara Amerika Serikat ke-49Masa jabatan3 Juli 1945 – 21 Januari 1947PresidenHarry S. Truman PendahuluEdward R. Stettinius, Jr.PenggantiGeorge C. MarshallDirektur Pimpinan Mobilisasi Perang ke-1Masa jabatan27 Mei 1943 – 3 Juli 1945sebagai Direktur ...

Artikel ini sebatang kara, artinya tidak ada artikel lain yang memiliki pranala balik ke halaman ini.Bantulah menambah pranala ke artikel ini dari artikel yang berhubungan atau coba peralatan pencari pranala.Tag ini diberikan pada November 2022. ロボジーRobo-GPoster layar lebarSutradaraShinobu YaguchiDitulis olehShinobu YaguchiPemeranMickey CurtisPenata musikMikki YoshinoSinematograferShinobu YaguchiPenyuntingRyuji MiyajimaPerusahaanproduksiFuji TelevisionAltamira PicturesDistributo...
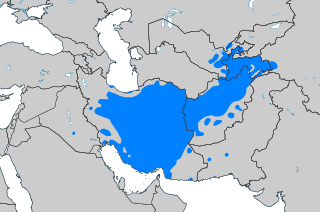
Bahasa Persia Baru فارسی نو Persia Modern, Dari Fārsi ditulis dalam Abjad Persia gaya Nastaliq Dituturkan diIran[1] (sebagai Farsi)Afganistan[1] (sebagai Dari)Tajikistan[1] (sebagai Tajik)Uzbekistan (sebagai Tajik)Iraq[2]Turkmenistan[3] (sebagai Tajik)Rusia (sebagai Tat)[4][5]Azerbaijan[6]Penutur90 juta sebagai bahasa pertama[7](110 juta keseluruhan)[6] Rincian data penutur Jumlah penutur beserta (jik...

Ordine per Servizio MeritorioOrder for Meritorious Service SudafricaTipologiaOrdine cavalleresco statale Statuscessato Istituzione24 ottobre 1986 Primo capoPieter Willem Botha Cessazione2 dicembre 2002 Ultimo capoThabo Mbeki GradiOroArgento Ordine più altoOrdine della Stella del Sudafrica Ordine più bassoOrdine della Buona Speranza Nastro dell'Ordine dal 1986 al 1996 Nastro dell'Ordine dal 1996 al 2002 Modifica dati su Wikidata · Manuale L'Ordine per Servizio Meritorio è stato un ord...

فيغاس بروالشعارمعلومات عامةنوع برامج تحرير الفيديو برامج تحرير الصوت نظام التشغيل مايكروسوفت ويندوز المنصة دوت نت فريموورك النموذج المصدري حقوق التأليف والنشر محفوظة متوفر بلغات الإنجليزية — الفرنسية — الإسبانية — الألمانية المطور الأصلي Sonic Foundry (en) المطورون Sonic Foundry (e...

Disambiguazione – Se stai cercando l'omonimo film del 1941 di Mervyn LeRoy, vedi Sorvegliato speciale (film 1941). Sorvegliato specialeSylvester Stallone in una scena del filmTitolo originaleLock Up Lingua originaleinglese Paese di produzioneStati Uniti d'America Anno1989 Durata115 min Generedrammatico, thriller, azione RegiaJohn Flynn SoggettoHenry Rosenbaum, Richard Smith, Jeb Stuart SceneggiaturaHenry Rosenbaum, Richard Smith, Jeb Stuart ProduttoreLawrence Gordon, Charles Gordon...

ماريانو راخويMariano Rajoy (بالإسبانية: Mariano Rajoy) رئيس وزراء إسبانيا في المنصب21 ديسمبر 2011 – 1 يونيو 2018 العاهل فيليب السادس خوسيه لويس ثباتيرو بيدرو سانشيز زعيم المعارضة في المنصب17 أبريل 2004 – 21 ديسمبر 2011 رئيس الوزراء خوسيه لويس ثباتيرو خوسيه لويس ثباتيرو ألفريدو بيريث روبالك...

この記事には複数の問題があります。改善やノートページでの議論にご協力ください。 大言壮語的な記述になっています。(2023年12月) 広告・宣伝活動的であり、中立的な観点で書き直す必要があります。(2023年12月) 株式会社向新Mukashin Co., Ltd.種類 株式会社市場情報 非上場本社所在地 日本〒598-0046大阪府泉佐野市羽倉崎1-5-10設立 1953年(昭和28年)9月15日(創業1892�...

Stratified complex society For other uses, see Civilization (disambiguation). The ancient Sumerians of Mesopotamia were the oldest civilization in the world, beginning about 4000 BCE. Ancient Egypt provides an example of an early culture civilization.[1] A civilization (British English: civilisation) is any complex society characterized by the development of the state, social stratification, urbanization, and symbolic systems of communication beyond natural spoken language (namely, a ...

First-level administrative division Administrative countyCategoryCountyLocationEngland and Wales and IrelandCreated byLocal Government Act 1888Local Government (Ireland) Act 1898CreatedEngland and Wales 1889Ireland 1899Abolished byLocal Government (Boundaries) Act (Northern Ireland) 1971Local Government Act 1972Local Government Act 2001AbolishedNorthern Ireland 1973England and Wales 1974Republic of Ireland 2002GovernmentCounty councilSubdivisionsRural districtUrban districtMunicipal...

West-east street in Manhattan, New York For other uses, see 34th Street (disambiguation). 34th StreetFrom the west end of 34th Street, looking east (c. 2009)Maintained byNYCDOTLength2.0 mi (3.2 km)[1]LocationManhattan, New York CityPostal code10001, 10121, 10016Coordinates40°45′02″N 73°59′23″W / 40.7506°N 73.9896°W / 40.7506; -73.9896West end NY 9A / West Side Highway in Hudson YardsEast end FDR Drive in Kips BayN...

Si ce bandeau n'est plus pertinent, retirez-le. Cliquez ici pour en savoir plus. Cet article relatif à la religion doit être recyclé (mars 2019). Une réorganisation et une clarification du contenu paraissent nécessaires. Améliorez-le, discutez des points à améliorer ou précisez les sections à recycler en utilisant {{section à recycler}}. Christogramme entouré par la prière du cœur en roumain : Doamne Iisuse Hristoase, Fiul lui Dumnezeu, miluieşte-mă pe mine păcătosul, s...

Australian rugby league footballer Rhys WesserPersonal informationFull nameRhys Joseph WesserBorn (1979-03-31) 31 March 1979 (age 45)Rockhampton, Queensland, AustraliaPlaying informationHeight180 cm (5 ft 11 in)Weight86 kg (13 st 8 lb)PositionFullback, Wing Club Years Team Pld T G FG P 1998–08 Penrith Panthers 178 113 1 0 460 2009–11 South Sydney 41 16 0 0 64 Total 219 129 1 0 524 Representative Years Team Pld T G FG P 2004–06 Queensland 4 0 0...

Элеонора Кастильская исп. Leonor de Castilla англ. Eleanor of Castile Бронзовая эффигия Элеоноры Кастильской в Вестминстерском аббатстве работы Уильяма Торелла[англ.] Королева Англии 20 ноября 1272 — 28 ноября 1290 Предшественник Элеонора Прованская Преемник Маргарита Французская Г�...

2012 Total Nonstop Action Wrestling pay-per-view event Destination X (2012)Promotional poster for the event featuring Austin AriesPromotionTotal Nonstop Action WrestlingDateJuly 8, 2012CityOrlando, FloridaVenueImpact ZoneAttendance1,100[1]Tagline(s)I Am The Main EventPay-per-view chronology ← PreviousSlammiversary 10 Next →Hardcore Justice Destination X chronology ← Previous2011 Next →2013 The 2012 Destination X was a professional wrestling pay-per-view (PP...

Basketball team in Mykolaiv, UkraineMBC MykolaivLeaguesUkrainian Basketball SuperLeagueFounded1967; 57 years ago (1967)History List Spartak Nikolaev(1972–1977)NKI Nikolaev(1977–1994)SK Mykolaiv(1994–2000)MBC Mykolaiv(2000–present) Capacity2,000LocationMykolaiv, UkraineTeam colorsWhite, Navy, Sea Blue Websitembcmklv.com Home Away Logo used since 2000 until 2018. Municipal Basketball Club Mykolaiv (in Ukrainian: Муніципальний Баске�...

American Revolutionary War military formation American Revolutionary War Armed Forces United States Continental Army → Commander-in-Chief → Regional departments → Units (1775, 1776, 1777–1784) → Manual Continental Navy Continental Marines State forces → List of militia units → List of state navies → Maritime units Great Britain List of British units France List of French units Related topics List of battles Military leadership vte The Connecticut Line was a formation within th...





