Selenium tetrafluoride
Selenium tetrafluoride (SeF4) is an inorganic compound. It is a colourless liquid that reacts readily with water. It can be used as a fluorinating reagent in organic syntheses (fluorination of alcohols, carboxylic acids or carbonyl compounds) and has advantages over sulfur tetrafluoride in that milder conditions can be employed and it is a liquid rather than a gas. SynthesisThe first reported synthesis of selenium tetrafluoride was by Paul Lebeau in 1907, who treated selenium with fluorine:[1]
A synthesis involving more easily handled reagents entails the fluorination of selenium dioxide with sulfur tetrafluoride:[2]
An intermediate in this reaction is seleninyl fluoride (SeOF2). Other methods of preparation include fluorinating elemental selenium with chlorine trifluoride:
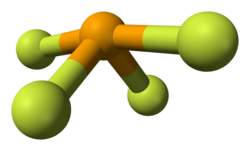
Structure and bondingSelenium in SeF4 has an oxidation state of +4. Its shape in the gaseous phase is similar to that of SF4, having a see-saw shape. VSEPR theory predicts a pseudo-trigonal pyramidal disposition of the five electron pairs around the selenium atom. The axial Se-F bonds are 177 pm with an F-Se-F bond angle of 169.2°. The two other fluorine atoms are attached by shorter bonds (168 pm), with an F-Se-F bond angle of 100.6°. In solution at low concentrations this monomeric structure predominates, but at higher concentrations evidence suggests weak association between SeF4 molecules leading to a distorted octahedral coordination around the selenium atom. In the solid the selenium center also has a distorted octahedral environment. ReactionsIn HF, SeF4 behaves as a weak base, weaker than sulfur tetrafluoride, SF4 (Kb= 2 X 10−2):
Ionic adducts containing the SeF3+ cation are formed with SbF5, AsF5, NbF5, TaF5, and BF3.[3] With caesium fluoride, CsF, the SeF5− anion is formed, which has a square pyramidal structure similar to the isoelectronic chlorine pentafluoride, ClF5 and bromine pentafluoride, BrF5.[4] With 1,1,3,3,5,5-hexamethylpiperidinium fluoride or 1,2-dimethylpropyltrimethylammonium fluoride, the SeF62− anion is formed. This has a distorted octahedral shape which contrasts to the regular octahedral shape of the analogous SeCl62−. [5] References
See alsoExternal links |
||||||||||||||||||||||||||||||||||||||||||||||||||||