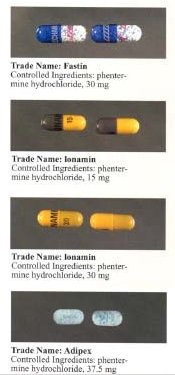
Phentermine
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Read other articles:

Mathematical proof that Euler's number (e) is irrational Part of a series of articles on themathematical constant e Properties Natural logarithm Exponential function Applications compound interest Euler's identity Euler's formula half-lives exponential growth and decay Defining e proof that e is irrational representations of e Lindemann–Weierstrass theorem People John Napier Leonhard Euler Related topics Schanuel's conjecture vte The number e was introduced by Jacob Bernoulli in 1683. More ...

artikel ini perlu dirapikan agar memenuhi standar Wikipedia. Tidak ada alasan yang diberikan. Silakan kembangkan artikel ini semampu Anda. Merapikan artikel dapat dilakukan dengan wikifikasi atau membagi artikel ke paragraf-paragraf. Jika sudah dirapikan, silakan hapus templat ini. (Pelajari cara dan kapan saatnya untuk menghapus pesan templat ini) Artikel ini tidak memiliki referensi atau sumber tepercaya sehingga isinya tidak bisa dipastikan. Tolong bantu perbaiki artikel ini dengan menamba...
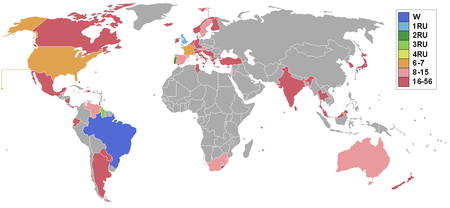
Miss World 1971 adalah kontes kecantikan Miss World ke-21 yang diselenggarakan tanggal 10 November 1971 di Royal Albert Hall London, Inggris.[1] 56 kontestan berkompetisi dalam ajang Miss World dan Lúcia Petterle dari Brazil meraih mahkota Miss World 1971.[2] She was crowned by Miss World 1970, Jennifer Hosten of Grenada. Hasil Countries and territories which sent delegates and results for Miss World 1971[2][3] Perolehan Hasil final Kontestan Miss World 1971 &...

American sailor and semi-legendary character (c. 1770/1780 – c. 1823) For other people named Mike Fink, see Mike Fink (disambiguation). This article uses bare URLs, which are uninformative and vulnerable to link rot. Please consider converting them to full citations to ensure the article remains verifiable and maintains a consistent citation style. Several templates and tools are available to assist in formatting, such as reFill (documentation) and Citation bot (documentation). (August 2022...

Grundordföljd är vilken ordning fraser kommer i en prototypisk huvudsats. Det rör sig alltså inte om egentlig ordföljd utan om vilken ordning satsdelarna subjekt (S), predikat (V) och objekt (O) kommer i. I allmänhet används termen verb i stället för predikat. De två vanligaste ordföljderna är SVO och SOV. Språk med grundordföljd med objektet före subjektet är mycket sällsynta. Lista över grundordföljder Dessa är de sex grundordföljder som kan bildas som permutationer av ...

American politician Allen M. FletcherAllen M. Fletcher54th Governor of VermontIn officeOctober 3, 1912 – January 7, 1915LieutenantFrank E. HowePreceded byJohn A. MeadSucceeded byCharles W. GatesMember of the Vermont Senate from Windsor CountyIn office1904–1905Serving with Edgar J. FishJames C. TaylorPreceded byLorenzo M. GreeneCharles H. HowardCharles DownerSucceeded byOliver W. DaleyFred L. DavisCharles F. AldrichMember of the Vermont House of Representatives from Ca...

Questa voce o sezione sull'argomento edizioni di competizioni calcistiche non cita le fonti necessarie o quelle presenti sono insufficienti. Puoi migliorare questa voce aggiungendo citazioni da fonti attendibili secondo le linee guida sull'uso delle fonti. Segui i suggerimenti del progetto di riferimento. Questa voce sull'argomento stagioni delle società calcistiche italiane è solo un abbozzo. Contribuisci a migliorarla secondo le convenzioni di Wikipedia. Segui i suggerimenti de...

Imperatori illiricidal 268 al 284L'Impero romano degli imperatori “legittimi” al centro, con l'Impero delle Gallie ad Occidente, il Regno di Palmira a Oriente, all'apice del periodo dell'Anarchia militare, poco dopo la morte di Gallieno (268), all'avvento dei cosiddetti Imperatori illirici PredecessoreGallieno SuccessoreTetrarchia Gli Imperatori illirici furono una serie di Imperatori che governarono l'Impero romano tra il 268 ed il 284. Questo nome deriva dalla provenienza geografica del...

Stroke resulting from lack of blood flow Medical conditionCerebral infarctCT scan slice of the brain showing a right-hemispheric cerebral infarct (left side of image).SpecialtyNeurology Cerebral infarction, also known as an ischemic stroke, is the pathologic process that results in an area of necrotic tissue in the brain (cerebral infarct).[1] In mid to high income countries, a stroke is the main reason for disability among people and the 2nd cause of death.[2] It is caused by...

First Maharaja of the Sikh Empire (1780–1839) For other uses, see Ranjit Singh (disambiguation). Sher-e-Punjab redirects here. For the hockey team, see Sher-e-Punjab (field hockey team). For the radio station, see KRPI. For the Doordarshan television series, see Maharaja Ranjit Singh (TV series). Ranjit SinghMaharaja of PunjabMaharaja of LahoreSher-e-Punjab (Lion of Punjab)Sher-e-Hind (Lion of India)Sarkar-i-Wallah (Head of Government)[1]Sarkar Khalsaji (Respected Head of the Khalsa...
Main pageTalkEmbassyRequested ArticlesMembersPortalRecognized contentTo doHelp Welcome to the discussion page of WikiProject United States Archives: Index 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15 Old U.S. notice board archives: National, Southern, Northern This page has archives. Sections older than 60 days may be automatically archived by Lowercase sigmabot III when more than 4 sections are present. Looking for Wikipedia talk:WikiProject United States in the Spanish Wikipedia? See ...

Italian prelate His EminenceFrancesco MontenegroCardinalArchbishop Emeritus of AgrigentoCardinal Francesco Montenegro during Consistory 2015.ChurchRoman Catholic ChurchArchdioceseAgrigentoSeeAgrigentoAppointed23 February 2008Installed17 May 2008Term ended22 May 2021PredecessorCarmelo FerraroSuccessorAlessandro DamianoOther post(s)Cardinal-Priest of Santi Andrea a Gregorio al Monte Celio (2015-)OrdersOrdination8 August 1969by Francesco FasolaConsecration18 March 2000by Giovanni Marra...

Halaman ini berisi artikel tentang komponis (1859-1924). Untuk hematologis (1927-2002), lihat Victor Herbert (hematologis). Victor Herbert Victor August Herbert (1 Februari 1859 – 26 Mei 1924) adalah seorang komponis, pemain cello dan konduktor Amerika yang dilahirkan di Irlandia dan dibesarkan di Jerman. Meskipun Herbert menikmati karier berpengaruh sebagai solois cello dan konduktor, ia dikenal karena mengkomposisikan beberapa operetta sukses yang tampil perdana di Broadway ...

Motorway in Scotland This article needs additional citations for verification. Please help improve this article by adding citations to reliable sources. Unsourced material may be challenged and removed.Find sources: M77 motorway – news · newspapers · books · scholar · JSTOR (March 2013) (Learn how and when to remove this message) M77M77 west of Newton Mearns, with the since demolished Netherplace Dye Works on the right.Route informationLength20.0 ...

Подводный вулкан Подводный вулкан — разновидность вулканов. Эти вулканы расположены на дне океана. Большинство современных вулканов расположено в пределах трёх основных вулканических поясов: Тихоокеанского, Средиземноморско-Индонезийского и Атлантического. Как с�...
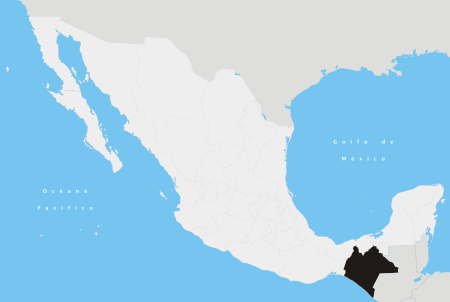
Place in ChiapasLas MargaritasLas MargaritasShow map of ChiapasLas MargaritasShow map of MexicoCoordinates: 16°18′55″N 91°58′54″W / 16.31528°N 91.98167°W / 16.31528; -91.98167Country Mexico (de jure) Rebel Zapatista Autonomous Municipalities (Controlled by)StateChiapasMunicipalityLas MargaritasVillage status9 December 1871City status24 March 1981Area • Municipality3,015 km2 (1,164 sq mi) • City10.08 km2 (3.89...

American chemist For other people named John D. Roberts, see J. D. Roberts (disambiguation) John D. RobertsJohn D. Roberts in 2010BornJohn Dombrowski Roberts(1918-06-08)June 8, 1918Los Angeles, California, U.S.DiedOctober 29, 2016(2016-10-29) (aged 98)Pasadena, California, U.S.NationalityAmericanAlma materUCLAAwards ACS Award in Pure Chemistry (1954) Roger Adams Award in Organic Chemistry (1967) William H. Nichols Medal (1972) Tolman Award (1974) Willard Gibbs Award (1983) Priestley...

Artikel ini bukan mengenai Kemiri. Stasiun Kemiri Bangunan baru Stasiun Kemiri, 2019LokasiKemiri, Kebakkramat, Karanganyar, Jawa Tengah 57762IndonesiaKoordinat7°31′48″S 110°54′8″E / 7.53000°S 110.90222°E / -7.53000; 110.90222Koordinat: 7°31′48″S 110°54′8″E / 7.53000°S 110.90222°E / -7.53000; 110.90222Ketinggian+98 mOperator Kereta Api IndonesiaDaerah Operasi VI Yogyakarta Letakkm 251+670 lintas Surabaya Kota-Kertosono-Mad...

Japanese manga series Machimaho: I Messed Up and Made the Wrong Person Into a Magical Girl!Cover of the first volume間違った子を魔法少女にしてしまった(Machigatta Ko o Mahō Shōjo ni Shiteshimatta)GenreMagical girl[1]Parody[2]Yankī[1] MangaWritten bySouryu [ja]Published byShinchoshaEnglish publisherNA: Seven Seas EntertainmentImprintBunch ComicsMagazineKurage BunchDemographicSeinenOriginal runOctober 7, 2016 – presentVolumes12...

Alessandro Tartagni Alessandro Tartagni, latinizzato come Alexander de Tartagnis e conosciuto anche come Alessandro da Imola (Imola, 1424 – 1477), è stato un giurista italiano, soprannominato nella sua epoca doctor aureus e doctor veritatis. Codicis partem commentaria, 1570 (Fondazione Mansutti, Milano). Indice 1 Biografia 2 Opere 2.1 Manoscritti 3 Note 4 Bibliografia 5 Altri progetti 6 Collegamenti esterni Biografia Studiò filosofia e diritto all'Università di Bologna, dove fu forse all...