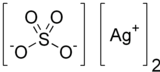Silver sulfate
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Read other articles:

World Miss UniversityTanggal pendirian1986TipeKontes kecantikanKantor pusatNew York CityLokasiAmerika SerikatBahasa resmi Bahasa InggrisPresidenSeung Min LeeSitus webhttp://wmu.world/ World Miss University (Korea: 월드 미스 유니버시티code: ko is deprecated ) adalah kontes kecantikan Internasional yang diadakan setiap tahun di Seoul, Korea Selatan sejak 1986,[1] dengan rata-rata sekitar 70 kontestan setiap tahun.[2] Acara ini bertujuan untuk memilih delegasi World Univ...

العلاقات الجزائرية السودانية الجزائر السودان تعديل مصدري - تعديل العلاقات الجزائرية السودانية تشيرإلى العلاقة التاريخية والحالية بين دولتين عربيتين أفريقيتين، الجزائر والسودان. الجزائر لديها سفارة في الخرطوم في حين أن السودان لديه سفارة في الجزائر. ال�...

Cet article est une ébauche concernant l’environnement et le Rwanda. Vous pouvez partager vos connaissances en l’améliorant (comment ?) selon les recommandations des projets correspondants. Consultez la liste des tâches à accomplir en page de discussion. L'environnement au Rwanda est l'environnement (ensemble des éléments - biotiques ou abiotiques - qui entourent un individu ou une espèce et dont certains contribuent directement à subvenir à ses besoins) du pays Rwanda,...

Fourth CrusadePart of the CrusadesA 15th-century miniature depicting the conquest of Constantinople by the Crusaders in 1204Date1202–1204LocationBalkans, Anatolia, DalmatiaResult Crusader victoryBelligerents Crusaders from: Kingdom of France County of Blois County of Brienne County of Champagne County of Flanders County of Saint-Pol Holy Roman Empire March of Montferrat County of Hainaut Republic of Venice In Europe: Byzantine Empire Kingdom of Hungary Kingdom of Croatia Holy Land: Ay...

تعداد المسلمين في خريطة العالم بالنسبة المئوية في كل بلد، وفقا لمنتدى بيو. (تقديرات 29 يونيو 2014). جزء من سلسلة مقالات حولالإسلام العقيدة الإيمان توحيد الله الإيمان بالملائكة الإيمان بالكتب السماوية الإيمان بالرسل والأنبياء الإيمان باليوم الآخر الإيمان بالقضاء والقدر أ�...

French ballet dancer Jean-Baptiste Landé. Jean-Baptiste Landé (died 26 February 1748) was a French ballet dancer, active in Sweden, Denmark and Russia. He is the founder of the Russian Ballet Mariinsky Ballet.[1] Sweden Landé was employed at the Polish royal court in Dresden when he was engaged by King Frederick I of Sweden in 1721. He was appointed dancing master of the Swedish court and in 1723 he became the director of the French Opera-Theatre in Bollhuset in Stockholm, which he...

Persikat KetapangNama lengkapPersatuan Sepak Bola Indonesia KetapangJulukanLaskar TanjungpuraBerdiri30 November 1987; 36 tahun lalu (1987-11-30)StadionStadion Tentemak Kabupaten Ketapang, Kalimantan Barat, Indonesia(Kapasitas: 20.000)PemilikAskab PSSI KetapangPelatihEddie Sibuea[1]LigaLiga 3 Regional Kalimantan Barat20191st (Juara) Kostum kandang Kostum tandang Persikat Ketapang atau Persatuan Sepakbola Indonesia Ketapang adalah klub sepak bola amatir Indonesia yang berbasis di K...

Diplomatic mission of the Swiss Confederation to the United States Embassy of Switzerland, Washington, D.C.LocationWashington, D.C.Address2900 Cathedral Avenue, N.W.Coordinates38°55′42″N 77°3′28″W / 38.92833°N 77.05778°W / 38.92833; -77.05778AmbassadorJacques Pitteloud The Embassy of Switzerland in Washington, D.C. (German: Schweizerische Botschaft in den Vereinigten Staaten, French: Ambassade de Suisse aux Etats-Unis d’Amérique, Italian: Ambasciata di S...

Gustav UcickyGustav Ucicky (1930)Lahir(1899-07-06)6 Juli 1899Wina, Austria-HungariaMeninggal27 April 1961(1961-04-27) (umur 61)Hamburg, Jerman BaratPekerjaanSutradaraTahun aktif1916–1961 Gustav Ucicky (6 Juli 1899 – 27 April 1961)[1] adalah seorang sutradara, penulis naskah dan sinemtografer asal Austria. Ia adalah salah satu sutradara paling sukses di Austria dan Jerman dari 1930an sampai awal 1960an.[2] Karyanya meliputi berbagai genre, namun...

For other battles at the same location, see Second Battle of Petrozsény and Third Battle of Petrozsény. First Battle of PetrozsényPart of the Battle of Transylvania of the Romanian Campaign of World War ICourse of the battle from 17 to 19 SeptemberDate17 – 22 September 1916LocationPetrozsény and the surrounding area, Transylvania, Austria-Hungary (today Petroșani, Romania)Result German victoryBelligerents Romania German EmpireCommanders and leaders Ioan Culcer Ioan Muică&#...

Science and Religion redirects here. For the 1991 book by John Hedley Brooke, see Science and Religion: Some Historical Perspectives. Science and theology redirects here. For the 1998 book by John Polkinghorne, see Science and Theology. God the Geometer — Gothic frontispiece of the Bible moralisée, representing God's act of Creation. France, mid-13th century The relationship between religion and science involves discussions that interconnect the study of the natural world, history, philos...

British puppeteer The topic of this article may not meet Wikipedia's notability guideline for biographies. Please help to demonstrate the notability of the topic by citing reliable secondary sources that are independent of the topic and provide significant coverage of it beyond a mere trivial mention. If notability cannot be shown, the article is likely to be merged, redirected, or deleted.Find sources: David Barclay puppeteer – news · newspapers · books ...

Wulan AyuningrumLahir14 Maret 1985 (umur 39) Jakarta, IndonesiaPekerjaanPebasket Wulan Ayuningrum (lahir 24 Maret 1985) adalah seorang pemain bola basket wanita Indonesia yang bertanding di WNBL Indonesia dengan memperkuat tim Tomang Sakti Mighty Bees Jakarta. Lulusan Perbanas Jakarta ini merupakan pemain bola basket wanita premier di Indonesia dengan telah membela negara dan menjadi anggota Tim Nasional Indonesia semenjak tahun 2001. Karier 2012, 2013, 2014 – WNBL Indonesia First Tea...

This article needs additional citations for verification. Please help improve this article by adding citations to reliable sources. Unsourced material may be challenged and removed.Find sources: Ramman festival – news · newspapers · books · scholar · JSTOR (December 2021) (Learn how and when to remove this message) Ramman performance Ramman is a religious festival and ritual theatre of the Garhwal region in India. It is a festival of the Garhwali ...

Artikel ini perlu diterjemahkan dari bahasa Inggris ke bahasa Indonesia. Artikel ini ditulis atau diterjemahkan secara buruk dari Wikipedia bahasa Inggris. Jika halaman ini ditujukan untuk komunitas bahasa Inggris, halaman itu harus dikontribusikan ke Wikipedia bahasa Inggris. Lihat daftar bahasa Wikipedia. Artikel yang tidak diterjemahkan dapat dihapus secara cepat sesuai kriteria A2. Jika Anda ingin memeriksa artikel ini, Anda boleh menggunakan mesin penerjemah. Namun ingat, mohon tidak men...

Полиция Боснии и Герцеговинысокращённо: Полиция БиГ босн. и хорв. Policija Bosne i Hercegovineсерб. Полициjа Босне и Херцеговине Общая информация Страна Босния и Герцеговина Юрисдикция Босния и Герцеговина Дата создания 1992 (в современном виде - 1997 год) Предшественник Милиция...

RRI SamarindaLPP RRI Stasiun SamarindaKotaSamarinda, Kalimantan TimurWilayah siarSeluruh wilayah Provinsi Kalimantan TimurFrekuensi 97.6 FM (Pro 1) 88.5 FM (Pro 2) 93.3 FM (Pro 3) 98.4 FM (Pro 4) Mulai mengudara20 Mei 1954; 70 tahun lalu (1954-05-20)FormatLihat Radio Republik Indonesia#RadioBahasaBahasa IndonesiaBahasa daerah di Kalimantan TimurOtoritas perizinanKementerian Komunikasi dan Informatika RIJaringanRRIPemilikLPP RRISitus webrri.co.id/samarinda Radio Republik Indonesia Samarin...
关于与「昆玉市」標題相近或相同的条目页,請見「昆玉」。 昆玉市قۇرۇمقاش شەھىرى县级市昆玉市 昆玉市在新疆的位置昆玉市被和田地區包圍昆玉市的地理位置坐标:37°12′34″N 79°17′27″E / 37.20944°N 79.29083°E / 37.20944; 79.29083国家 中华人民共和国隶属行政区新疆维吾尔自治区新疆生产建设兵团第十四师語源崑岡出美玉[1]政府�...

Government body in the United States For the laws that this administers, see Copyright law of the United States and Law of the United States. United States Copyright OfficeAgency overviewFormedJuly 8, 1870; 154 years ago (1870-07-08)JurisdictionFederal government of the United StatesHeadquartersWashington, D.C.Agency executiveShira Perlmutter, Register of CopyrightsParent agencyLibrary of CongressWebsitecopyright.gov The James Madison Memorial Building, which houses the offi...

FIFA Ballon d'or 2011 Lionel Messi.Généralités Sport Football Organisateur(s) FIFA et France Football Éditions 56e Catégorie Trophée mondial Date 2011 Participants 23 joueurs nommés Palmarès Vainqueur Lionel Messi (3) Deuxième Cristiano Ronaldo Troisième Xavi Navigation Édition précédente Édition suivante modifier Le FIFA Ballon d'or 2011 est un trophée récompensant le meilleur footballeur du monde au cours de l'année civile 2011. La 56e remise du trophée du Ballon d'o...