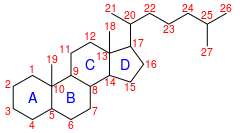
Cholestane
| |||||||||||||||||||||||||||||||||||||||||||||
Read other articles:

Sjarifuddin Baharsjah Menteri Pertanian Indonesia ke-20Masa jabatan16 Maret 1993 – 14 Maret 1998PresidenSoeharto PendahuluWardojoPenggantiJustika Baharsjah Informasi pribadiLahir(1932-05-16)16 Mei 1932Sindanglaut, Lemahabang, Keresidenan Cirebon, Hindia BelandaMeninggal15 Januari 2021(2021-01-15) (umur 88)Jakarta, IndonesiaKebangsaanIndonesiaSuami/istriJustika Baharsjah (m. 1962)HubunganLeila Chairani Budiman (saudara)Anak2Orang tuaSutan Baha...

Boonie Bears: Mystical WinterNama lain熊出没之雪岭熊风SutradaraDing LiangLiu FuyuanPerusahaanproduksiShenzhen Huaqiang Shuzi Dongman Co.,LtdLe Vision Pictures (Tianjin) Co.,LtdFantawild Holdings IncPearl River Pictures Co., LtdBeijing Iqiyi Co.,LtdBeijing LeTV Mobile Media & Technology Co. LtdHe Yi information technology (Beijing) Co., LTDTencent VideoTV.SOHU.COMYou Yang( Tian Jin) Dong Man Culture Media Co., LTDDistributorLe Vision Pictures (Tianjin) Co.,LtdMr. Cartoon ...

William BucklandPotret Buckland karya Samuel Cousins (1833)Lahir(1784-03-12)12 Maret 1784Axminster, Devon, InggrisMeninggal14 Agustus 1856(1856-08-14) (umur 72)Islip, Oxfordshire,[1] InggrisKebangsaanInggrisAlmamaterWinchester CollegeCorpus Christi CollegeDikenal atasMegalosaurus, koprolitSuami/istriMary Buckland (m. invalid year)PenghargaanCopley Medal (1822)Wollaston Medal (1848)Karier ilmiahBidangPalaeontologi William Buckland DD, FRS (12 Maret ...

Карта Украины. Список заказников, расположенных на территории Украины. Заказники, располагающиеся на территории де-факто входящего в состав России Крымского полуострова[1] вынесены в отдельный список. Содержание 1 А 2 Б 3 В 4 Г 5 Д 6 З 7 К 8 Л 9 Н 10 О 11 П 12 Р 13 С 14 Т 15 У 16 Ф 17 Х 18 Ч...

Untuk sutradara Korea pada masa sebelumnya dengan nama yang sama, lihat Kim Ki-duk (kelahiran 1934). Ini adalah nama Korea; marganya adalah Kim. Kim Ki-dukNama KoreaHangul김기덕 Hanja金基德 Alih AksaraGim GideokMcCune–ReischauerKim Kidŏk Kim Ki-duk (Hangul: 김기덕 Pengucapan Korea: [kimɡidʌk]; kelahiran 20 Desember 1960) adalah seorang pembuat film Korea Selatan yang dikenal karena karya-karya layar lebari art-house idiosinkratik buatannya. Film-filmnya meraih bebe...

فلوتونفيل الإحداثيات 42°56′48″N 74°22′10″W / 42.9467°N 74.3694°W / 42.9467; -74.3694 [1] تقسيم إداري البلد الولايات المتحدة[2] التقسيم الأعلى مقاطعة مونتغومري خصائص جغرافية المساحة 1.345879 كيلومتر مربع1.346274 كيلومتر مربع (1 أبريل 2010) ارتفاع 88 متر عدد السك�...

Brain blood supply Cerebral circulationAreas of the brain are supplied by different arteries. The major systems are divided into an anterior circulation (the anterior cerebral artery and middle cerebral artery) and a posterior circulationSchematic of veins and venous spaces that drain deoxygenated blood from the brainIdentifiersMeSHD002560Anatomical terminology[edit on Wikidata] Cerebral circulation is the movement of blood through a network of cerebral arteries and veins supplying the br...

Species of bat Northern broad-nosed bat Conservation status Least Concern (IUCN 3.1)[1] Scientific classification Domain: Eukaryota Kingdom: Animalia Phylum: Chordata Class: Mammalia Order: Chiroptera Family: Vespertilionidae Genus: Scotorepens Species: S. sanborni Binomial name Scotorepens sanborni(Troughton, 1937)[2] The northern broad-nosed bat (Scotorepens sanborni) is a species of the vespertilionid family of microbats. It can be found in northern Australia, Ti...

Cet article est une ébauche concernant une chanson, le Concours Eurovision de la chanson et la Russie. Vous pouvez partager vos connaissances en l’améliorant (comment ?) selon les recommandations des projets correspondants. You Are the Only One Sergueï Lazarev interprétant You Are the Only One durant une répétition, avant la première demi-finale du Concours Eurovision de la chanson 2016. Chanson de Sergueï Lazarev au Concours Eurovision de la chanson 2...

Chronologies Chronologie du sport 2006 2007 2008 2009 2010 2011 2012Mois :Jan - Fév - Mar - Avr - Mai - Juin Juil - Aoû - Sep - Oct - Nov - Déc 2008 ◄◄ 2009 en sport ►► 2010 Chronologie dans le monde 2006 2007 2008 2009 2010 2011 2012Décennies :1970 1980 1990 2000 2010 2020 2030Siècles :XIXe XXe XXIe XXIIe XXIIIeMillénaires :Ier IIe IIIe Chronologies géographiques Afrique Afrique du Sud, Algérie, ...

Indian politician Shiv PrakashJoint National General Secretary (Organisation), Bharatiya Janata PartyIncumbentAssumed office July 2014President Amit Shah J. P. Nadda Personal detailsBorn (1967-08-01) 1 August 1967 (age 56)NationalityIndianPolitical partyBharatiya Janata PartyAlma materMahatma Jyotiba Phule Rohilkhand UniversityOccupationPoliticianWebsitewww.bjp.org/hi/national-general-secretarys Shiv Prakash (born 1 August 1967) is an Indian politician and the current Joint National ...

Ice hockey team in Pittsburgh, PennsylvaniaPittsburgh BankersCityPittsburgh, PennsylvaniaLeagueWestern Pennsylvania Hockey LeagueOperated1899–1904, 1907–1909Home arenaDuquesne GardenColorsVariousOwner(s)Various local banksChampionshipsRegular season titles(2) 1902–03, 1907–08 The Pittsburgh Bankers were one of the earliest professional ice hockey clubs. The club was based in Pittsburgh, Pennsylvania and was a member of the Western Pennsylvania Hockey League, the first league to openly...

American missionary Lorenzo LyonsLorenzo Lyons circa 1880Born(1807-04-18)April 18, 1807Colrain, MassachusettsDiedOctober 6, 1886(1886-10-06) (aged 79)Waimea, Hawaii County, HawaiiOther namesMakua LaianaKnown forMissionary to HawaiiSpouses Betsy Curtis Lucia Smith His grave site. Lorenzo Lyons or Makua Laiana (April 18, 1807 – October 6, 1886) was an early missionary to the Kingdom of Hawaii. He was a songwriter who wrote the lyrics of Hawaiʻi Aloha, which was inducted into t...

† Стеллерова корова Муляж стеллеровой коровы в Лондонском музее естествознания Научная классификация Домен:ЭукариотыЦарство:ЖивотныеПодцарство:ЭуметазоиБез ранга:Двусторонне-симметричныеБез ранга:ВторичноротыеТип:ХордовыеПодтип:ПозвоночныеИнфратип:Челюстно�...

Pour le film, voir Romulus et Rémus (film). Pour les articles homonymes, voir Romulus (homonymie). Romulus Romulus et Rémus (sculpture de la fontaine de la place du Capitole). Titre 1er roi légendaire de Rome (en partie avec Titus Tatius) 753 – 716 av. J.-C.(37 ans[1]) Avec Titus Tatius Prédécesseur Aucun Successeur Numa Pompilius Biographie Titre complet Fondateur de Rome, roi Date de naissance 771 av. J.-C.[1] (Albe la Longue) Date de décès 716 av. J.-C. (Rome) Nationalité L...

Australian coastal city This article is about the coastal city in Australia. For the local government area, see City of Gold Coast. Gold CoastQueenslandQ1 and the Gold Coast skylineBroadbeachIsle of Capri CanalsLight Rail on Surfers Paradise BoulevardChevron RenaissanceHome of the ArtsRoxy Theatre at Warner Bros. Movie WorldGold CoastCoordinates28°01′0″S 153°24′0″E / 28.01667°S 153.40000°E / -28.01667; 153.40000Population640,778 (2021)[1][2...

This article needs additional citations for verification. Please help improve this article by adding citations to reliable sources. Unsourced material may be challenged and removed.Find sources: King's College of Engineering – news · newspapers · books · scholar · JSTOR (June 2018) (Learn how and when to remove this message) Engineering college in IndiaKings College of Engineeringகிங்ஸ் பொறியியல் கல்லூரிLocat...

TikalKuil I Tikal dengan ketinggian yang mencapai 47 m[1]Nama alternatifYax MutalLokasiFlores, Departemen Petén, GuatemalaWilayahCekungan PeténKoordinat17°13′19″N 89°37′25″W / 17.22194°N 89.62361°W / 17.22194; -89.62361Koordinat: 17°13′19″N 89°37′25″W / 17.22194°N 89.62361°W / 17.22194; -89.62361SejarahPeriodeKlasik Awal hingga Klasik AkhirBudayaPeradaban Maya Situs Warisan Dunia UNESCONama resmi: Tama...

Allied tactical operation of World War II Operation CottagePart of the Aleutian Islands campaign of World War IIAmerican troops landing on KiskaDateAugust 15, 1943LocationKiska, Aleutian Islands, Alaska, United StatesResult Allied victoryBelligerents United States Canada Japan (not present)Commanders and leaders Charles Corlett Harry W. Foster Not presentUnits involved 7th Infantry Division 4th Infantry Regiment 53rd Infantry Regiment 184th Infantry Regiment 10th Mountain Div...

Passione d'amoreFosca (Valeria D'Obici)Titolo originalePassione d'amore Paese di produzioneItalia, Francia Anno1981 Durata117 min Generedrammatico RegiaEttore Scola SoggettoIginio Ugo Tarchetti (romanzo), Ettore Scola, Ruggero Maccari SceneggiaturaEttore Scola, Ruggero Maccari ProduttoreFranco Committeri FotografiaClaudio Ragona MontaggioRaimondo Crociani MusicheArmando Trovajoli ScenografiaFiorenzo Senese Interpreti e personaggi Valeria D'Obici: Fosca Bernard Giraudeau: Giorgio Bacchetti...