Chain reaction
|
Read other articles:

Stasiun Kawane-Koyama川根小山駅Stasiun Kawane-Koyama pada Juli 2009LokasiOkuizumi, Kawanehon-cho, Haibara-gun, Shizuoka-kenJepangKoordinat35°08′30.41″N 138°08′27.90″E / 35.1417806°N 138.1410833°E / 35.1417806; 138.1410833Koordinat: 35°08′30.41″N 138°08′27.90″E / 35.1417806°N 138.1410833°E / 35.1417806; 138.1410833Ketinggian354 meterOperator Ōigawa RailwayJalur■ Jalur IkawaLetak5.8 kilometer dari SenzuJumlah peron2...

Tauge gorengTauge goreng kakilimaNama lainTaoge goreng, toge gorengSajianutama atau kudapanTempat asalIndonesiaDaerahJakarta dan Bogor, Jawa baratSuhu penyajianpanasBahan utamatauge (kecambah kacang hijau) ditumis dengan air panas, ditambah irisan ketupat, tahu, dan mi, disajikan dengan kuah oncom.Sunting kotak info • L • BBantuan penggunaan templat ini Media: Tauge goreng Tauge goreng adalah hidangan vegetarian Indonesia yang bercitarasa gurih yang dibuat dengan cara ...

Evanescence EPAlbum mini karya EvanescenceDirilisDecember 1998 (Little Rock, Arkansas)Direkam1997GenreAlternative RockDurasi29:18LabelPrivate releaseProduserBen Moody, Jason JonesKronologi Evanescence —String Module Error: Match not foundString Module Error: Match not found Evanescence EP(1998) Sound Asleep EP(1999)Sound Asleep EP1999 Evanescence EP adalah EP pertama dari grup band rock Evanescence. EP ini dirilis oleh Amy Lee dan Ben Moody pada sebuah konser pada tahun 1998. CD-R-nya m...

本條目存在以下問題,請協助改善本條目或在討論頁針對議題發表看法。 此條目需要补充更多来源。 (2018年3月17日)请协助補充多方面可靠来源以改善这篇条目,无法查证的内容可能會因為异议提出而被移除。致使用者:请搜索一下条目的标题(来源搜索:羅生門 (電影) — 网页、新闻、书籍、学术、图像),以检查网络上是否存在该主题的更多可靠来源(判定指引)。 �...

منتخب البوسنة والهرسك لكأس ديفيز البلد البوسنة والهرسك الكابتن أمير ديلتش تصنيف ITF 27 كأس ديفيز أول سنة 1996 سنوات اللعب 24 Ties played (W–L) 93 (51–42) سنوات فيمجموعة العالم 0 (0–0) أكثر إجمالي إنتصارات ميرزا باشيتش (27–17) تعديل مصدري - تعديل منتخب البوسنة والهرسك لكأس ديفيز هو ممثل...

Artikel ini sebatang kara, artinya tidak ada artikel lain yang memiliki pranala balik ke halaman ini.Bantulah menambah pranala ke artikel ini dari artikel yang berhubungan atau coba peralatan pencari pranala.Tag ini diberikan pada Desember 2023. Kumiko OhbaNama asal大場 久美子Lahir6 Januari 1960 (umur 64)Kawaguchi, Saitama, JapanKebangsaanJepangPendidikanHatogaya Municipal Nakai Elementary SchoolHatogaya Municipal Hachimangi Junior High SchoolTokyo Seitoku College University Hi...

Wakil Bupati MeranginPetahanaTidak adasejak 22 September 2023Masa jabatan5 tahunDibentuk2003Pejabat pertamaDrs. H. Ubay AliSitus webmeranginkab.go.id Berikut ini adalah daftar Wakil Bupati Merangin dari masa ke masa. No Potret Wakil Bupati Mulai Jabatan Akhir Jabatan Prd. Ket. Bupati 1 Drs. H.Ubay Ali 2003 2008 1 H.Rotani YutakaS.H. 2 Drs. H.Hasan Basri Harun 2008 2013 2 Kompol (Purn.) Dr. Drs. H.NalimS.H., M.M. 3 Drs. H.Abdul KhafidM.M. 6 Agustus 2013 15 Februari 2018 3 &#...

1995 film by Barbet Schroeder This article is missing information about the film's production, and theatrical/home media releases. Please expand the article to include this information. Further details may exist on the talk page. (July 2019) Kiss of DeathPromotional film posterDirected byBarbet SchroederScreenplay byRichard PriceStory byEleazar LipskyBased onKiss of Death1947 filmby Ben HechtCharles LedererProduced bySusan Hoffman Barbet SchroederStarring David Caruso Kathryn Erbe Helen Hunt ...

История Грузииსაქართველოს ისტორია Доисторическая Грузия Шулавери-шомутепинская культураКуро-араксская культураТриалетская культураКолхидская культураКобанская культураДиаухиМушки Древняя история КолхидаАриан-КартлиИберийское царство ФарнавазидыГруз�...

1993 promotional single by CeCe PenistonSearchin'Promotional single by CeCe Penistonfrom the album Thought 'Ya Knew A-side I'm in the Mood I'm Not Over You ReleasedDecember 7, 1993 (1993-12-07)Recorded1993Genre House dance Length3:43 (Album version)LabelA&MSongwriter(s) Steve Hurley Tonia Hurley M-Doc Producer(s)Steve Silk Hurley Searchin' is a 1993 song by the musician CeCe Peniston, taken from her second solo album Thought 'Ya Knew on A&M Records. However, the...
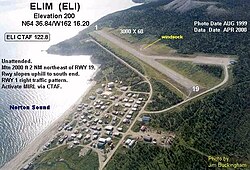
AirportElim AirportIATA: ELIICAO: PFELFAA LID: ELISummaryAirport typePublicOwnerState of Alaska DOT&PF - Northern RegionServesElim, AlaskaElevation AMSL162 ft / 49 mCoordinates64°36′54″N 162°16′14″W / 64.61500°N 162.27056°W / 64.61500; -162.27056MapELILocation of airport in AlaskaRunways Direction Length Surface ft m 1/19 3,401 1,037 Gravel Statistics (2018)Based aircraft (2018)0Passengers4,298Freight732,000 lbsSource: Federal Aviation Ad...

John Turturro al Tribeca Film Festival 2017 John Michael Turturro (New York, 28 febbraio 1957) è un attore, regista e sceneggiatore statunitense con cittadinanza italiana. Indice 1 Biografia 2 Filmografia 2.1 Attore 2.1.1 Cinema 2.1.2 Televisione 2.2 Doppiatore 2.3 Regista 2.4 Sceneggiatore 3 Riconoscimenti 4 Doppiatori italiani 5 Onorificenze 6 Note 7 Bibliografia 8 Altri progetti 9 Collegamenti esterni Biografia Turturro nasce a Brooklyn, borough di New York, il 28 febbraio del 1957, figli...

CSKA Sofia 2010–11 football seasonCSKA Sofia2010–11 seasonChairmanDimitar BorisovManager Pavel Dochev (until August 16) Gjore Jovanovski (until October 21) Milen Radukanov (from October 21)A Group Third placeBulgarian CupWinnersUEFA Europa LeagueGroup StageTop goalscorerLeague: Delev (13)All: Delev (22)Highest home attendance12,300 vs Levski Sofia (1 August 2010)Lowest home attendance450 vs Kaliakra (17 October 2010) Home colours Away colours ← 2009–102011–12 → T...

Paolo Bertini Informazioni personali Arbitro di Calcio Sezione Arezzo Professione Promotore finanziario Attività nazionale Anni Campionato Ruolo 1998-2008 Serie A e B Arbitro Attività internazionale 2003-2008 UEFA e FIFA Arbitro Paolo Bertini (Arezzo, 7 luglio 1964) è un ex arbitro di calcio italiano. Indice 1 Biografia 1.1 Vicende giudiziarie 2 Dopo il ritiro 3 Note 4 Bibliografia Biografia Avvicinatosi all'arbitraggio seguendo le orme paterne[1], fu inserito nella CAN su segnala...

Part of a series onForced labour and Slavery Contemporary Child labour Child soldiers Conscription Debt Forced marriage Bride buying Child marriage Wife selling Forced prostitution Human trafficking Peonage Penal labour Contemporary Africa 21st-century jihadism Sexual slavery Wage slavery Historical Antiquity Egypt Babylonia Greece Rome Medieval Europe Ancillae Black Sea slave trade Byzantine Empire Kholop Prague slave trade Serfs History In Russia Emancipation Thrall Venetian slave trade Ba...

Israel's official memorial to the Jewish victims of the Holocaust Yad Vashemיָד וַשֵׁםAerial view of Yad VashemEstablished19 August 1953LocationOn the western slope of Mount Herzl, also known as the Mount of Remembrance, a height in western Jerusalem, IsraelCoordinates31°46′27″N 35°10′32″E / 31.77417°N 35.17556°E / 31.77417; 35.17556TypeIsrael's official memorial to the victims of the HolocaustVisitorsabout 925,000 (2017),[1] 800,000...

Pour les articles homonymes, voir Nothomb. Cet article est une ébauche concernant la Belgique, le Luxembourg et un cours d’eau. Vous pouvez partager vos connaissances en l’améliorant (comment ?) selon les recommandations des projets correspondants. Nothomb(lb) Noutemerbaach Caractéristiques Longueur 9 km Bassin collecteur Rhin Cours Source Holtz Confluence Attert · Localisation Grendel · Coordonnées 49° 45′ 09″ N, 5° 48′ 48″ E Géog...

Novel by Australian writer Morris West The Big Story AuthorMorris WestLanguageEnglishGenreFictionPublisherHeinemannPublication date1957Publication placeAustraliaMedia typePrintPages227pp.Preceded byKundu Followed byThe Second Victory The Big Story (1957) is a novel by Australian writer Morris West.[1] It was also published in the USA under the title The Crooked Road.[1] The book was adapted into a movie under that title. Frontispiece quotation It must...

Questa voce o sezione sugli argomenti storia e LGBT è priva o carente di note e riferimenti bibliografici puntuali. Sebbene vi siano una bibliografia e/o dei collegamenti esterni, manca la contestualizzazione delle fonti con note a piè di pagina o altri riferimenti precisi che indichino puntualmente la provenienza delle informazioni. Puoi migliorare questa voce citando le fonti più precisamente. Segui i suggerimenti dei progetti di riferimento 1, 2. La neutralità di questa voce o se...

City in Minnesota, United States City in Minnesota, United StatesAda, MinnesotaCityNorman County CourthouseCongregational Church of AdaAda Village HallNickname: Heart of the ValleyMotto(s): Alive and Thriving!Location of Adawithin Norman County and state of MinnesotaCoordinates: 47°17′55″N 96°30′57″W / 47.29861°N 96.51583°W / 47.29861; -96.51583CountryUnited StatesStateMinnesotaCountyNormanFounded1874IncorporatedFebruary 9, 1881Government •...
![{\displaystyle (1)...{\frac {d{\ce {[CH4]}}}{dt}}=k_{2}{\ce {[CH3]}}{\ce {[CH3CHO]}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/d61b20e140c0c1867ce20d407e63e4a382f22efd)
![{\displaystyle (2)...{\frac {d{\ce {[CH_3]}}}{dt}}=k_{1}{\ce {[CH3CHO]}}-k_{2}{\ce {[CH3]}}{\ce {[CH3CHO]}}+k_{3}{\ce {[CH3CO]}}-2k_{4}{\ce {[CH3]}}^{2}=0}](https://wikimedia.org/api/rest_v1/media/math/render/svg/c4fad9080a897fc119c3267a037fd3e619f4e8e5)
![{\displaystyle (3)...{\frac {d{\ce {[CH3CO]}}}{dt}}=k_{2}{\ce {[CH3]}}{\ce {[CH3CHO]}}-k_{3}{\ce {[CH3CO]}}=0}](https://wikimedia.org/api/rest_v1/media/math/render/svg/6f352bc089e9bd504a0a15d2866fcdbe16ec7d1d)
![{\displaystyle k_{1}{\ce {[CH3CHO]}}-2k_{4}{\ce {[CH3]}}^{2}=0}](https://wikimedia.org/api/rest_v1/media/math/render/svg/26e20a3084b86d2eb7c68dd8e905fddfee39afa9)
![{\displaystyle (4)...{\ce {[CH3]}}={\frac {k_{1}}{2k_{4}}}{\ce {[CH3CHO]}}^{1/2}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/d710bb49bc6630e4e3ce83f893fa49dac25740f6)
![{\displaystyle (5){\frac {d{\ce {[CH4]}}}{dt}}={\frac {k_{1}}{2k_{4}}}k_{2}{\ce {[CH3CHO]}}^{3/2}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/b60bf787fb9c9d686fb84f3e5ec43b87ff10d60c)