Phorbol
| |||||||||||||||||||||||||||||||||||||||||||
Read other articles:

Lokasi Kabupaten Maluku Barat Daya di Provinsi Maluku Berikut ini adalah daftar kecamatan, kelurahan, dan desa di Kabupaten Maluku Barat Daya, Provinsi Maluku, Indonesia. Kabupaten Maluku Barat Daya terdiri atas 17 kecamatan, 1 kelurahan, dan 117 desa dengan luas wilayah 4.581,06 km² dan jumlah penduduk 66.805 jiwa (2017). Kode Wilayah Kabupaten Maluku Barat Daya adalah 81.07.[1][2][3][4] Kode Wilayah Nama Kecamatan Ibu kota Jumlah Daftar Kelurahan dan Desa Ke...

Flüchtlingsandrang vor dem Notaufnahmelager Marienfelde in Berlin, Juli 1961 Flucht aus der Sowjetischen Besatzungszone und der DDR – im Sprachgebrauch der DDR „Republikflucht“ – war das Verlassen der DDR oder ihres Vorläufers, der Sowjetischen Besatzungszone (SBZ), oder Ost-Berlins ohne Genehmigung der Behörden. Von der Gründung der DDR am 7. Oktober 1949 bis in den Juni 1990 verließen über 3,8 Millionen Menschen den Staat, davon viele illegal und unter großer Gefahr...
Ini adalah nama Korea; marganya adalah Na. Na Ah-reumNa Ah-reum (2018)Informasi pribadiLahir24 Maret 1990 (umur 34)Provinsi Jeolla Utara, Korea Selatan Nama KoreaHangul나아름 Hanja羅雅凜 Alih AksaraNa A-reumMcCune–ReischauerNa Arŭm Na Ah-reumInformasi pribadiCatatan membalapDisiplinSepeda balapan jalan rayaTrekPosisiRider Na Ah-reum (Pengucapan Korea: [na.a.ɾɯm]) adalah pebalap sepeda putri asal Korea Selatan, yang lahir di Naju.[1][2] Pada Pesta Olahraga ...

Men's basketball of Santa Clara University Santa Clara Broncos 2023–24 Santa Clara Broncos men's basketball team UniversitySanta Clara UniversityFirst season1904All-time record1,418-1,019 (.582)Head coachHerb Sendek (8th season)ConferenceWest Coast ConferenceLocationSanta Clara, CaliforniaArenaLeavey Center (Capacity: 4,500)NicknameBroncosColorsMaroon and white[1] Uniforms Home Away Alternate NCAA tournament Final Four1952NCAA tournament Elite Eight1952, 1...

Film and television production company TriStar ProductionsCompany typeJoint ventureIndustryFilmFoundedAugust 9, 2013; 10 years ago (2013-08-09)[1]FounderThomas Rothman[2]Headquarters10202 West Washington Boulevard, Culver City, California, United States[3]Key peopleThomas Rothman (Chairman)Lindsay Sloane (SVP, TV)David Beaubaire (EVP, film)Nicole Brown (SVP, film)[3]ProductsMotion picturesTelevision productionOwnerSony Pictures EntertainmentTh...

Calendaring and mail server Microsoft Exchange ServerExchange Server 2019 logoDeveloper(s)MicrosoftInitial releaseApril 2, 1996; 28 years ago (1996-04-02)[1]Stable release2019 CU14 Mar24SU (15.02.1544.009)[2] (March 12, 2024; 45 days ago (2024-03-12)) [±] Operating systemWindows ServerPlatformx64TypeCollaborative softwareLicenseProprietary commercial softwareWebsitewww.microsoft.com/en-us/microsoft-365/exchange/email Micro...

This article needs additional citations for verification. Please help improve this article by adding citations to reliable sources. Unsourced material may be challenged and removed.Find sources: List of submarine-borne aircraft – news · newspapers · books · scholar · JSTOR (February 2024) (Learn how and when to remove this message) A seaplane launching from British submarine HMS M2. HMS M2 retrieving her seaplane This is a list of aircraft carried und...

Rajnath Singh Menteri Urusan Dalam NegeriPetahanaMulai menjabat 26 Mei 2014Perdana MenteriNarendra ModiPendahuluSushilkumar ShindePenggantiPetahanaPresiden Partai Bharatiya JanataMasa jabatan23 Januari 2013 – 8 Juli 2014PendahuluNitin GadkariPenggantiAmit ShahMasa jabatan24 Desember 2005 – 24 Desember 2009PendahuluL K AdvaniPenggantiNitin GadkariKetua Menteri Uttar PradeshMasa jabatan28 Oktober 2000 – 8 Maret 2002GubernurSuraj BhanVishnu Kant ShastriPendah...
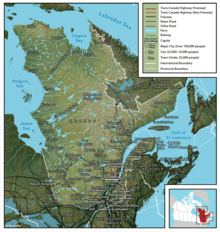
Region of Eastern Canada Not to be confused with Canadian Prairies. PlaceCentral Canada Centre du Canada (French)Map of Central Canada, defined politicallyCoordinates: 50°N 79°W / 50°N 79°W / 50; -79CompositionOntarioQuebecLargest cityTorontoLargest metroGreater Toronto AreaCanadian Confederation1 July 1867Area (2021 - land,[1] 2017 - fresh water[2][3]) • Total2,552,291.51 km2 (985,445.26 sq mi) •&...

Species of bird Wood thrush Song Song Conservation status Least Concern (IUCN 3.1)[1] Scientific classification Domain: Eukaryota Kingdom: Animalia Phylum: Chordata Class: Aves Order: Passeriformes Family: Turdidae Genus: HylocichlaBaird, 1864 Species: H. mustelina Binomial name Hylocichla mustelina(Gmelin, JF, 1789) Breeding Migration Nonbreeding Synonyms[2] Turdus mustelinus Gmelin, 1789 Merula mustelina (Gmelin, 1789) Catharus mu...

2016年美國總統選舉 ← 2012 2016年11月8日 2020 → 538個選舉人團席位獲勝需270票民意調查投票率55.7%[1][2] ▲ 0.8 % 获提名人 唐納·川普 希拉莉·克林頓 政党 共和黨 民主党 家鄉州 紐約州 紐約州 竞选搭档 迈克·彭斯 蒂姆·凱恩 选举人票 304[3][4][註 1] 227[5] 胜出州/省 30 + 緬-2 20 + DC 民選得票 62,984,828[6] 65,853,514[6]...
2020年夏季奥林匹克运动会波兰代表團波兰国旗IOC編碼POLNOC波蘭奧林匹克委員會網站olimpijski.pl(英文)(波兰文)2020年夏季奥林匹克运动会(東京)2021年7月23日至8月8日(受2019冠状病毒病疫情影响推迟,但仍保留原定名称)運動員206參賽項目24个大项旗手开幕式:帕维尔·科热尼奥夫斯基(游泳)和马娅·沃什乔夫斯卡(自行车)[1]闭幕式:卡罗利娜·纳亚(皮划艇)&#...

stasiun Namioka浪岡駅Stasiun Namioka pada Juli 2010Lokasi61 Hosoda, Namioka, Aomori-shi, Aomori-ken 038-1311JepangKoordinat40°42′38.11″N 140°34′52.22″E / 40.7105861°N 140.5811722°E / 40.7105861; 140.5811722Koordinat: 40°42′38.11″N 140°34′52.22″E / 40.7105861°N 140.5811722°E / 40.7105861; 140.5811722Operator JR EastJalur■ Jalur Utama ŌuLetak462.1 km dari FukushimaJumlah peron1 peron samping + 1 peron pulauInformasi l...

Canadian ice hockey player (born 1997) Ice hockey player Alex Barré-Boulet Barré-Boulet with the Syracuse Crunch in 2020Born (1997-05-21) May 21, 1997 (age 27)Montmagny, Quebec, CanadaHeight 5 ft 10 in (178 cm)Weight 170 lb (77 kg; 12 st 2 lb)Position CentreShoots LeftNHL teamFormer teams Montreal CanadiensTampa Bay LightningSeattle KrakenNHL draft UndraftedPlaying career 2018–present Alex Barré-Boulet (born May 21, 1997) is a Canadian professi...

Protected area in New South Wales, AustraliaMutawintji National ParkNew South WalesIUCN category II (national park) Aboriginal rock art located within the national park, 1976.Mutawintji National ParkNearest town or cityWhite CliffsCoordinates31°08′48″S 142°22′53″E / 31.14667°S 142.38139°E / -31.14667; 142.38139Established4 September 1998 (1998-09-04)[1]Area689.12 km2 (266.1 sq mi)[1]Managing authoritiesNSW Nati...

Cette page concerne l'année 910 du calendrier julien. Pour l'année 910 av. J.-C., voir 910 av. J.-C. Pour le nombre 910, voir 910 (nombre). Chronologies La péninsule ibérique en 910.Données clés 907 908 909 910 911 912 913Décennies :880 890 900 910 920 930 940Siècles :VIIIe IXe Xe XIe XIIeMillénaires :-IIe -Ier Ier IIe IIIe Calendriers Romain Chinois Grégorien Julien Hébraïque Hindou Hégirien Persan Républicain modi...

New Zealand politician For the association footballer, see William Anderton. The HonourableBill Anderton16th Minister of Internal AffairsIn office12 December 1957 – 12 December 1960Prime MinisterWalter NashPreceded bySidney Walter SmithSucceeded byLeon GötzMember of the New Zealand Parliamentfor EdenIn office27 November 1935 – 27 November 1946Preceded byArthur StallworthySucceeded byWilfred FortuneMember of the New Zealand Parliamentfor Auckland CentralIn office27 Novem...

De facto executive government in France (1793–1794) For other uses, see Committee of Public Safety (disambiguation). Committee of Public SafetyComité de salut public (French)TypeProvisional governmentStatusDisestablishedAppointerNational ConventionConstituting instrumentNational ConventionFormation6 April 1793Abolished25 October 1795SuccessionExecutive Directory The Committee of Public Safety (French: Comité de salut public) was a committee of the National Convention which formed the...

Consonantal sound represented by ⟨ŋ⟩ in IPA Velar nasal and Agma redirect here. For the voiceless consonant, see Voiceless velar nasal. For other uses, see Agma (disambiguation). Voiced velar nasalŋIPA Number119Audio sample source · helpEncodingEntity (decimal)ŋUnicode (hex)U+014BX-SAMPANBraille Image The voiced velar nasal, also known as eng, engma, or agma (from Greek ἆγμα âgma 'fragment'), is a type of consonantal sound used in some spoken languages. It i...
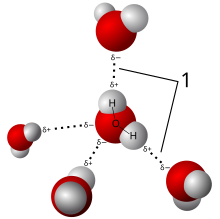
Intermolecular attraction between a hydrogen-donor pair and an acceptor Model of hydrogen bonds (1) between molecules of water AFM image of naphthalenetetracarboxylic diimide molecules on silver-terminated silicon, interacting via hydrogen bonding, taken at 77 K.[1] (Hydrogen bonds in the top image are exaggerated by artifacts of the imaging technique.[2][3][4]) In chemistry, a hydrogen bond (or H-bond) is primarily an electrostatic force of attraction between ...


